Half of adalimumab patients did not give consent for biosimilar switch, survey reveals - The Pharmaceutical Journal

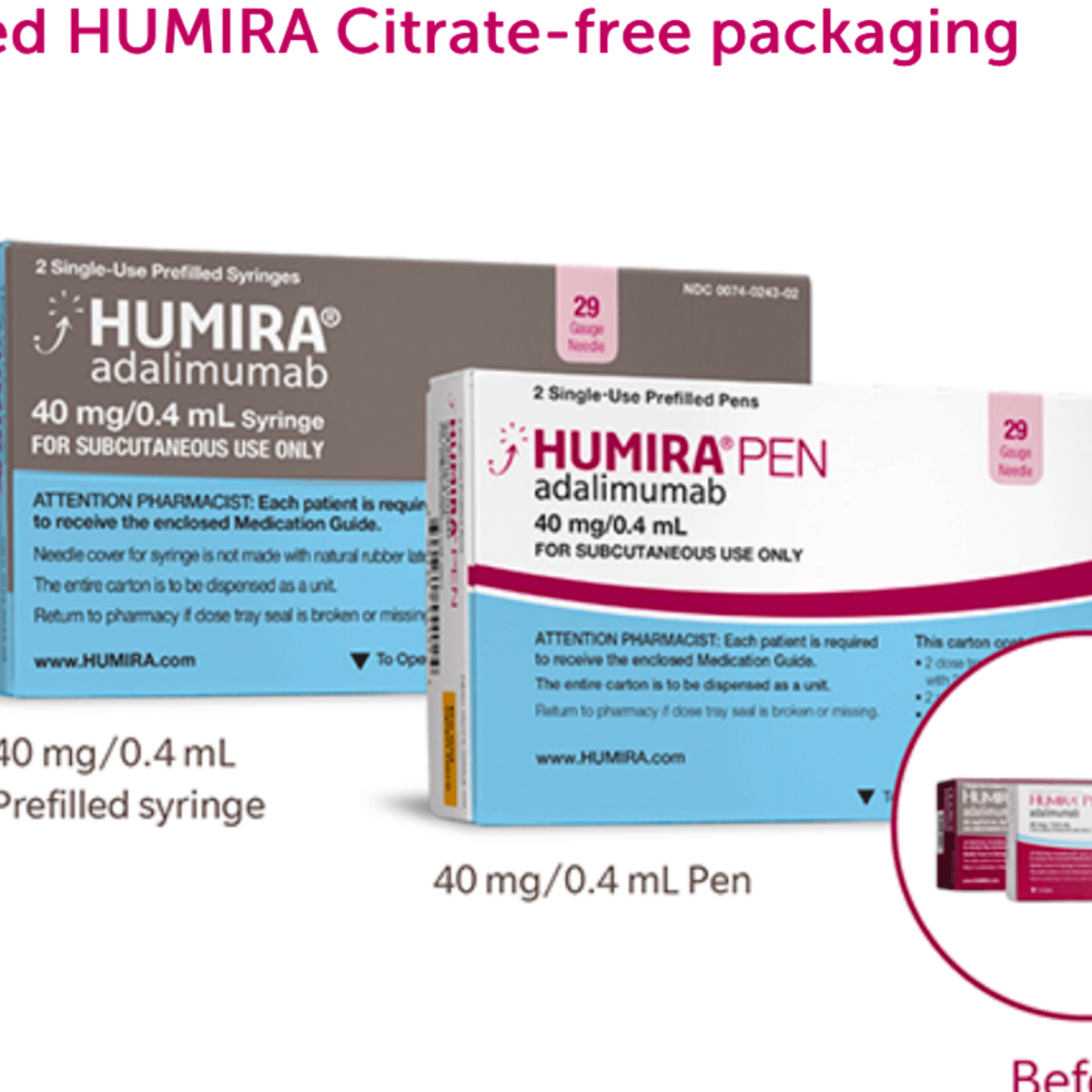
dtw Research, Inc on Twitter: "Humira Introduces New Citrate-Free Formulation in the Q1 2019 Biologics and Non-Biologics for GI Indications @abbvie #dtw #marketHighlights #abbvie #Humira #citratefree #adalimumab #GI #GIIndications #Q12019 ...
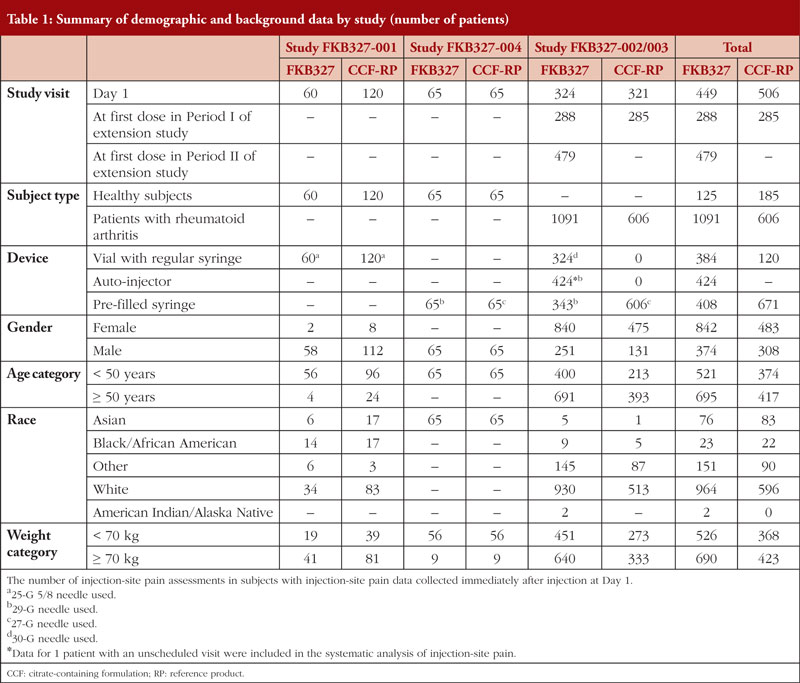



:max_bytes(150000):strip_icc()/iStock-186131854-59540d905f9b5815d91d1360.jpg)